Top 10 Host Cell Protein ELISA Applications You Should Know?
In the realm of biopharmaceutical development, the determination of host cell proteins (HCPs) is vital. Dr. Emily Dawson, a leading expert in protein analytics, emphasizes, "Understanding host cell protein ELISA is crucial for ensuring product safety." The accurate quantification of HCPs helps mitigate potential immunogenicity in therapeutic proteins.
Host cell protein ELISA applications are numerous and diverse. Companies use them to assess the purity of biopharmaceuticals. While the methodologies have progressed, some challenges remain. The sensitivity and specificity of assays can vary, leading to inconsistent results. This variability calls for continuous evaluation and improvement of existing ELISA techniques.
Moreover, regulatory guidelines around HCP levels are ever-evolving. It is essential for researchers to remain updated on the latest recommendations. As we delve into the top ten applications of host cell protein ELISA, we will explore their significance and practical implications. Despite advancements, there is always room for enhanced techniques and accuracy in HCP detection.

Understanding Host Cell Proteins and Their Importance in Bioprocessing
Host cell proteins (HCPs) are contaminants found in biopharmaceutical products. They originate from the organism used for producing proteins, often in cell culture processes. Understanding HCPs is crucial in bioprocessing because they can affect product quality and safety. The presence of HCPs can elicit immune responses in patients, leading to adverse effects. Manufacturers must measure these proteins to ensure the safety of therapeutic proteins.
To effectively manage HCP levels, various assay techniques are employed, such as ELISA. These assays provide quantitative data on HCP levels, enabling manufacturers to assess process performance. The development of HCP assays requires an understanding of antibody specificity. Testing must be rigorous, with protocols reviewed and validated. A single misstep in assay development can lead to inaccurate results, impacting patient safety. Regular updates based on evolving knowledge and technology are essential for maintaining reliability.
The landscape of HCP management is continually evolving, and learning from past experiences is vital. Each development stage brings challenges. Innovating without losing sight of safety is a delicate balance. Investing in education and research builds competence in this field, ensuring progress while protecting patient health.
Top 10 Host Cell Protein ELISA Applications You Should Know
| Application | Description | Importance |
|---|---|---|
| Release Testing | Evaluating the levels of host cell proteins in biopharmaceuticals before release. | Ensures product safety and efficacy. |
| Process Development | Monitoring HCP levels during different stages of bioprocessing. | Optimizes purification processes. |
| Stability Studies | Assessing the impact of storage conditions on HCP levels. | Ensures product stability over time. |
| Comparative Analysis | Comparing HCP profiles between different production runs. | Identifies variations that could affect quality. |
| Regulatory Compliance | Ensuring that HCP levels meet regulatory requirements. | Facilitates the approval process for new drugs. |
| Downstream Processing | Monitoring HCPs after purification steps. | Enhances efficiency of purification methods. |
| Adventitious Agent Testing | Assessing HCP levels as part of pathogen safety evaluations. | Minimizes risks in biologics manufacturing. |
| Clinical Trials | Monitoring HCP levels in clinical trial materials. | Ensures safety for trial participants. |
| Product Characterization | Characterizing HCP profiles in relation to product function. | Understanding effects on biological activity. |
| Method Development | Developing new ELISA methods for HCP detection. | Improves specificity and sensitivity in assays. |
Overview of ELISA Technology for Host Cell Protein Detection
ELISA, or enzyme-linked immunosorbent assay, is a crucial technology for detecting host cell proteins (HCPs). HCPs are inevitable impurities in recombinant protein production. Their presence can impact product safety and efficacy. ELISA provides a reliable method for quantifying these proteins, ensuring that biopharmaceuticals meet regulatory standards.
The principle of ELISA is straightforward. It uses antibodies to capture specific proteins from a sample. A secondary antibody, linked to an enzyme, is then applied. The enzyme reacts with a substrate to produce a measurable signal. This signal correlates with the amount of HCP present. However, developing a valid assay can be challenging. Factors like antibody specificity and detection limits require careful consideration.
Despite its advantages, ELISA is not without limitations. Cross-reactivity can lead to false positives. It can also miss certain HCPs if antibodies are not available. Continuous refinement of the assay is essential. Researchers must regularly evaluate their protocols to adapt to changing needs. Balancing sensitivity and specificity remains a persistent challenge in the field.
Top 10 Host Cell Protein ELISA Applications
Top Applications of ELISA for Monitoring Biopharmaceutical Production Processes
ELISA applications for monitoring biopharmaceutical production processes are essential in today's biotechnology landscape. According to industry reports, nearly 70% of biopharmaceutical products rely on protein quantification techniques to ensure safety and efficacy. Host cell proteins (HCPs) can affect product quality and immunogenicity, making their quantification critical during production.
One common ELISA application is real-time monitoring of HCP levels. This method allows for timely adjustments in the manufacturing process. For instance, if HCP levels exceed acceptable thresholds, corrective actions can be implemented immediately, reducing the risk of costly batch failures. Additionally, regulatory agencies have begun to emphasize HCP quantification as part of the quality control process, further validating the importance of these applications.
Tips: Implementing an ELISA method requires proper validation. Confirm sensitivity, specificity, and reproducibility through rigorous testing. It’s also vital to maintain accurate documentation throughout the process.
A reflective aspect of using ELISA is ensuring the assay is tailored to the specific HCPs in question. Generic assays may overlook unique proteins present in specific production systems. Custom assays may take longer to develop but provide essential insights into product safety. Striking a balance between time efficiency and comprehensive analysis is imperative for effective biopharmaceutical manufacturing.
Utilizing ELISA in Characterizing Host Cell Protein Variants
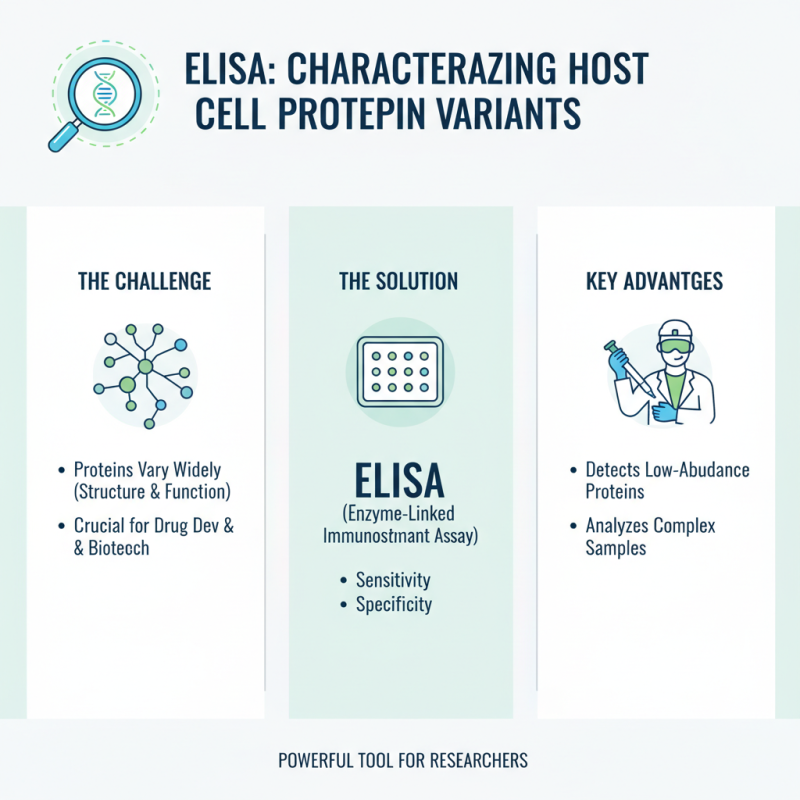
Utilizing ELISA in characterizing host cell protein variants offers a powerful tool for researchers. These proteins can vary widely in their structure and function. Identifying these differences is crucial for drug development and biotechnology applications. ELISA stands out due to its sensitivity and specificity. Researchers can leverage this method to detect low-abundance proteins in complex samples.
Tips: Ensure your samples are prepared correctly. Contaminants can lead to unreliable results. Consistency in sample handling is key.
Moreover, understanding the structural variations of host cell proteins can enhance therapeutic efficacy. Each variant may interact differently with the therapeutic proteins. ELISA can facilitate this analysis, allowing scientists to refine their products more effectively. Utilizing proper controls in experiments is essential for accurate interpretations.
Tips: Always run a standard curve for quantification accuracy. This ensures reliable comparisons between different assays. Regular calibration of equipment also helps maintain data integrity.
Compliance and Regulatory Considerations for Host Cell Protein ELISA Applications
Host Cell Protein (HCP) ELISA applications play a critical role in biopharmaceutical development. These techniques ensure product safety and efficacy by detecting residual host cell proteins that may remain after manufacturing. Regulatory agencies, such as the FDA and EMA, emphasize the need for robust HCP testing protocols. In fact, 75% of biopharmaceutical companies report regulatory compliance issues tied to inadequate HCP assessment, according to a recent industry report.
When developing HCP ELISA applications, consideration of regulatory guidelines is paramount. The International Council for Harmonisation (ICH) proposes stringent limits on HCP levels for biologics. Some reports indicate that non-compliance can lead to delays in product approval, hindering therapeutic availability. For instance, a lack of a comprehensive HCP strategy has caused up to 50% of clinical trials to experience setbacks.
Despite the progress in HCP quantification, challenges remain. The high variability of host cell protein profiles complicates the establishment of universal testing methods. Additionally, many labs struggle with the sensitivity and specificity of their ELISA assays. Continuous validation and improvement of these applications are essential to meet the evolving regulatory landscape and scientific expectations. Embracing best practices in HCP testing can significantly reduce risks and enhance overall product quality.
Related Posts
-

Why Are Host Cell Proteins Important in Bioprocessing?
-

How to Choose the Right Protein Expression Systems for Your Research?
-
Top Advances in Cell Gene Therapy and Their Impact on Medicine?
-

2026 Top Types of Growth Factors Shaping Future Industries?
-

2026 Top Methods in Protein Extraction for Enhanced Research Results?
-

Top 10 Human Cell Lines Used in China for Research and Development?
